
Project HDAB-LU
Preparing digital business capabilities for Health Data Access Body Luxembourg
HDAB-LU is a four-year project to prepare and lay the technical foundations for the future Health Data Access Body (HDAB) in Luxembourg. Kicked off in January 2024, the project is a cornerstone of the national implementation of the European Health Data Space (EHDS) regulation. The objective of HDAB-LU is to setup the digital business capabilities needed to unlock the potential of health data for a variety of secondary uses by policy-makers, scientists, innovators, and civil society. To achieve this objective, the project develops tools, services, and the necessary infrastructure to fulfil the requirements defined by the EHDS regulation related to data discoverability, accessibility, and reusability of health data in Luxembourg.
HDAB-LU is funded by the European Commission’s EU4Health Programme via a direct grant to Member States. Luxembourg National Data Service (LNDS) was nominated by the Ministry of Health and Social Security to apply for the grant and to coordinate the implementation of the project.
It is important to note that the HDAB-LU project is a preparatory project for the future HDAB. It is not the assignment of the HDAB role. The HDAB or HDABs will be formally designated by the new Luxembourgish legislation implementing the EHDS. When assigned, the outcome of the HDAB-LU project will be handed over to the future HDAB or HDABs of Luxembourg.
What is an HDAB?
The EHDS aims to create an EU-wide health data sharing framework. An essential building block of this are HDAB to be created by Member States. An HDAB’s primary mission is to facilitate data discovery and to orchestrate legitimate data access requests between data users and data holders. HDABs are also intended to facilitate data access and –exchanges of health data across the EU Member States.
In a nutshell, HDABs are thus one-stop platforms for data discovery (i.e. what data is available?), data access request management (i.e. how can I apply for access?), and secure data processing (i.e. how do I use data in a safe and privacy-preserving way?). All these features will be available in one interoperable Europe-wide network of HDABs linking to the European central service.
What does the HDAB-LU platform consist of?
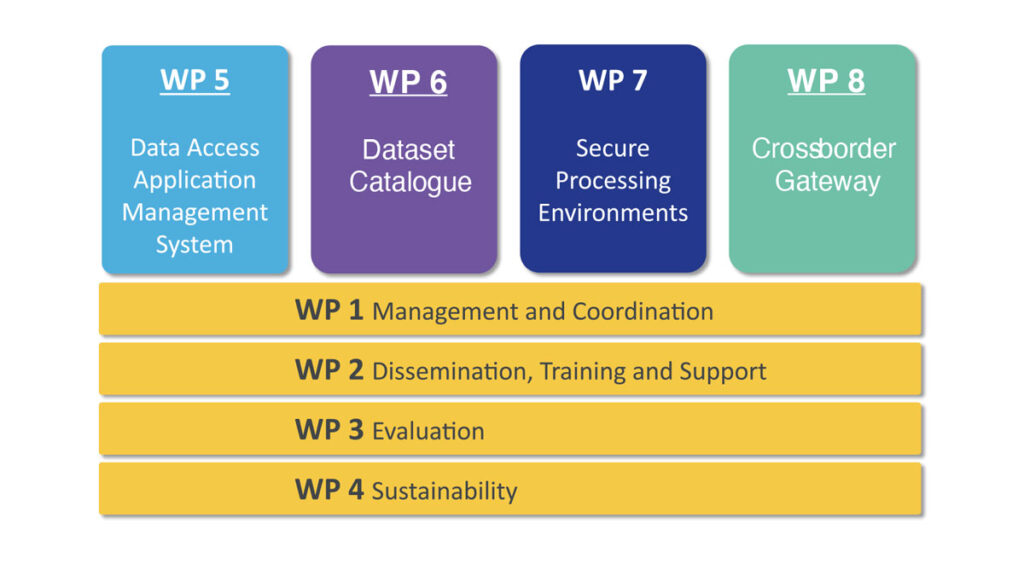
The HDAB-LU platform consists of four components, which are being developed under the four vertical work packages for the benefit of the future HDAB:
- WP5 – National Data Access Application management solution
- WP6 – National Dataset Catalogue for Health Data
- WP7 – Secure Processing Environments for Health Data
- WP8 – Cross-border gateway to connect with the HealthData@EU infrastructure
To enable data discovery, information about accessible health data (i.e. the metadata describing the health data) must first be documented, organised, and structured in the HDAB platform. The resulting, openly available metadata are then collected and listed in the HDAB’s data catalogue.
In addition to hosting the data catalogue, the HDAB also handles processing of data access requests. Data access requests submitted to the designated HDAB will be reviewed, approved, or denied by the authorities, based on rules and procedures defined in the EHDS regulation. If approved, the HDAB will issue and publish a “data permit” and provide access to a secure processing environment (SPE) in which data can be safely made accessible and analysed. As required by EHDS, HDABs will also establish gateways for cross-border applications and requests by building an interface to the EHDS central service at the EU level to provide metadata as well as to exchange data applications and requests.
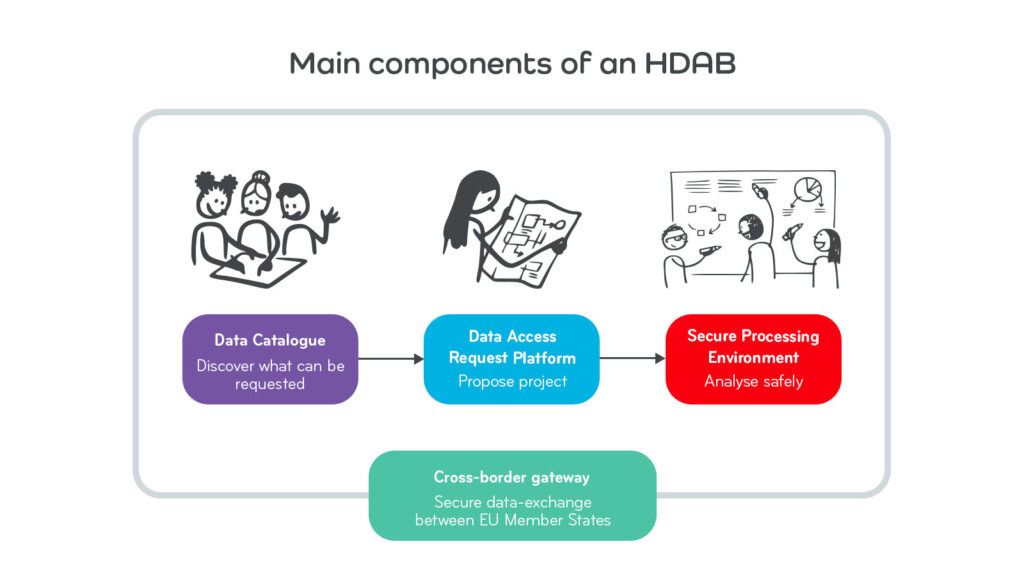
For the purpose of secondary use (e.g. for using health data for research, innovation or public policy) the HealthData@EU platform is being created by the European Commission. A key component of the HealthData@EU network within the EHDS is the “cross-border gateway”. This gateway will allow secure exchange of metadata and application-related information between national and European central service. Via this gateway, health data users (i.e. researchers) can submit health data access applications across EU Member States and to automatically forward the health data access applications to the relevant contact points. Hence, it gives researchers in the EU access to far more health data to conduct research.
The HDAB structure thus ensures that data can be reused in a privacy preserving way, assuring citizens that their data is safe. At the same time, it enables data reuse for purposes, such as discovery of new medicine, which are in the interest of society. This fosters trust, discovery, and innovation.
How is the HDAB-LU project structured?
The work plan for HDAB-LU is organised in two types of work packages: Horizontal work packages provide certain functions across the entire project, while vertical work packages develop the digital business capabilities in the scope of this project.
The horizontal work packages are:
WP1 – Management and Coordination
WP2 – Dissemination, training and support
WP3 – Evaluation
WP4 – Sustainability
The vertical work packages are:
WP5 – National Data Access Application management solution
WP6 – National Dataset Catalogue for Health Data
WP7 – Secure Processing Environments for Health Data
WP8 – Cross-border gateway to connect with the HealthData@EU infrastructure
What are the expected benefits of HDAB-LU?
HDAB-LU develops integrated services that will be needed for the future Health Data Access Body of Luxembourg. Through the HDAB-LU project, we increase the readiness of the future HDAB and ensure alignment with the standards and technical requirements with other HDABs.
How can I contact you?
An HDAB is in everyone’s interest. Therefore, the HDAB-LU project aims to shape the technical and organisational functionalities together with its stakeholders to create a successful model benefitting individuals and society.
Do you have questions or ideas for HDAB-LU? Please contact our project team at: info@lnds.lu
Glossary & footnotes
| Metadata | Information that describes the data. It doesn’t include the data itself. |
| Regulation | A “regulation” is a binding legislative act. It must be applied in its entirety across the EU. Source: European Union. |
| Secondary use of health data | When health data is reused for research, innovation, public health, policy-making and personalised medicine, in European law this is referred to as ‘secondary’ use. This data is used for example to identify trends, develop new treatments and improve healthcare services. Source: European Commission |
Disclaimer

Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or the European Health and Digital Executive Agency. Neither the European Union nor the granting authority can be held responsible for them.


